Product range
BASIC product line
Professional absorbable haemostats based on oxidized cellulose in the form of a sterile knitted fabric.
ADVANCED product line
Professional absorbable haemostats based on oxidized cellulose in the form of a sterile absorbable soft layered material.
Why to choose OKCEL®

PREMIUM COTTON, PREMIUM CARE
OKCEL® is a 100% natural material that is produced by selective oxidation of the finest extra-long staple cotton.

SUPERIOR HAEMOSTASIS
OKCEL® is the ultimate solution for quickly stopping bleeding. It is characterized by a high haemostatic capacity, which is positively influenced by the excellent absorption of the material and its large surface area.5 Haemostasis can be achieved in under 1.5 minutes.3,4

MAXIMUM PERFORMANCE, HIGH COMPATIBILITY
In the operating room, seconds matter – but so does the long-term result. OKCEL® is a haemostatic agent based on oxidized cellulose, known as a biodegradable, bioresorbable and biocompatible 1 polymer with no adverse effects on the body, which has been used in healthcare for decades. OKCEL® thus offers more than just effective bleeding control – it also ensures maximum biocompatibility with the human body, promoting rapid healing without negative effects on surrounding tissues. This cutting-edge, biologically degradable and bioresorbable solution offers the perfect blend of advanced technology and nature’s purity.

ABSORBABILITY WITHOUT RESIDUE, WITHOUT REACTION, WITHOUT WORRIES
OKCEL® is perfectly accepted by the body, typically being absorbed within 14 days with practically no tissue reaction and without residues, depending on the amount of product used, the degree of blood saturation, and the character of tissue.3, 4

FIGHTS BACTERIA, HELPS TISSUE
OKCEL® products have proven antimicrobial activity against a wide range of pathogens6 (including antibiotic-resistant bacteria MRSA, PRSP, VRE, MRSE), thereby also supporting subsequent tissue regeneration. The use of oxidized cellulose is therefore associated with significant advantages in many medical applications.7

BROAD PORTFOLIO, THE ONLY GOAL – A SAFE PROCEDURE
OKCEL® is indicated for the control of internal capillary, venous, and minor arterial bleeding during a wide range of surgical procedures, including minimally invasive procedures, where conventional haemostatic methods such as sutures or ligatures are not effective or feasible.2 The broad product portfolio allows you to select the right solution for your specific surgical procedure.

PERFECTLY UNDER CONTROL – EVEN DURING THE PROCEDURE
OKCEL® facilitates work in the surgical field due to its exceptional handling, re-positioning capability, and shape stability, ensuring precise and reliable placement where needed.5 The ideal choice for demanding surgical situations.

PACKAGING YOU CAN RELY ON
The packaging of OKCEL® products is designed to meet the strictest healthcare standards. It provides maximum product protection, prevents microbial contamination, and minimizes the risk of damage throughout the entire storage period. At the same time, the packaging is easy to open and intuitive to handle, which facilitates the work of medical personnel in critical moments.

A TRADITION THAT COMMITS
OKCEL® is manufactured by an established European company with a century-long history, and the production of oxidized cellulose itself is backed by three decades of experience.
1. For OKCEL ® products proven by biocompatibility testing in accordance with EN ISO 10993. Data on file: 18640-033.
2. Based on OKCEL® instructions for use. Data on file: 18640-033.
3.Jindřich Lahovský, MD. Evaluation of efficacy and safety of medical devices series OKCEL®. Study Report Ref. No AP-SY-1501. Data on file: 18640 – 033.
4. Jindřich Lahovský, MD. Evaluation of efficacy and safety of the medical device OKCEL® S. Study Report Ref. No AP-SY-1701. Data on file: 18640 – 033.
5. Jindřich Lahovský, MD. Závěrečná zpráva laboratorního vyhodnocení vlastností hemostatik na bázi oxidované regenerované a neregenerované celulózy. Study Report Ref. No. AP-SY-1901. Data on file: 18640 – 034.
6. Ing. Iveta Brožková, Ph. D. Department of Biological and Biochemical Sciences at the Faculty of Chemical Technology, University of Pardubice. Final report on testing the antimicrobial activity of the product OKCEL®. Data on file: 18640 - 034.
7. However, OKCEL® is not intended to replace therapeutic or prophylactic antimicrobial intervention with pharmaceuticals to control or prevent postoperative infections.
Mode of action
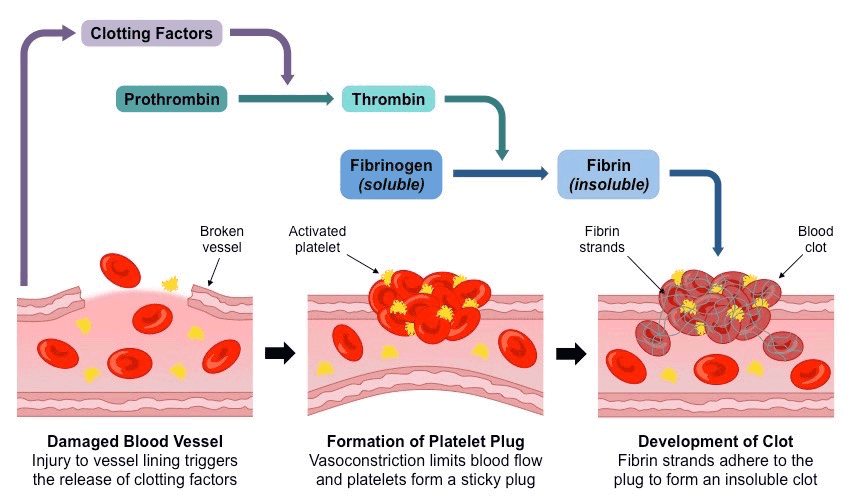
When saturated with blood, OKCEL® swells into a gelatinous mass, still maintaining its original structure. The product helps in the formation of a clot by initial denaturation of blood proteins, thus supporting local haemostasis and hemorrhage control. The activation of the coagulation system of blood plasma is initiated very likely by the negatively charged surface of the oxidized cellulose which results in thrombin generation and only subsequently in activation and adhesion of platelets and fibrin formation (soluble fibrinogen transforms into a net of insoluble fibrin, which stops the bleeding).1, 2
The process, by which blood clots are formed, involves a complex set of reactions collectively called the coagulation cascade:3
When used properly in minimal amounts, OKCEL® is absorbed with practically no tissue reaction, depending on the degree of saturation with blood, the quantity of product used and the character of tissue.
1. Kollár, P. et al. Hemostatické účinky oxidované celulosy. Česká a slovenská farmacie, 2008; 57, 11-16
2. Ryšavá, J. et al. Surface interactions of oxidized cellulose with fibrin(ogen) and blood platelets. Sensors and Actuators B: Chemical 90(1-3):243-249, April 2003. DOI: 10.1016/S0925-4005(03)00035-2.
3. https://ib.bioninja.com.au/standard-level/topic-6-human-physiology/63-defence-against-infectio/clotting.html


